Analysis of the world’s first asteroid gas sample that was returned by the Hayabusa2 mission has been published in “Science Advances”.
October 21, 2022 (JST)
Japan Aerospace Exploration Agency
Kyushu University
Ibaraki University
Tokyo Institute of Technology
Japan Agency for Marine-Earth Science and Technology
Hokkaido University
Tohoku University
Kyoto University
Hiroshima University
The University of Tokyo
The Japan Aerospace Exploration Agency (JAXA) is conducting the first analysis of the sample returned from asteroid Ryugu by the Hayabusa2 mission with the Hayabusa2 Initial Analysis Team, comprising of six sub-teams and two Phase-2 curation institutes. Results from the Initial Analysis Team’s “Volatile Component Analysis Team” have been published in the US Journal “Science Advances” on October 21, 2022 (JST).
Title:First asteroid gas sample delivered by the Hayabusa2 mission: A treasure box from Ryugu
The initial analysis of the asteroid Ryugu samples
The sample from asteroid Ryugu was returned to Earth by the Hayabusa2 spacecraft on December 6, 2020. The initial description of the sample (Phase-1 curation) followed as the facility established at JAXA’s Institute of Space and Astronautical Science (ISAS). Parts of the sample were then distributed to the Hayabusa2 Initial Analysis Team, which consists of six sub-teams and two Phase-2 curation institutes. This division into specialized sub-teams is designed to achieve the scientific objectives of Hayabusa2 through high-precision analysis of different aspects of the sample. Data from the Phase-2 curation institutes’ comprehensive analysis of the grain characteristics will be included in the JAXA Hayabusa2 grain catalogue for future analysis proposals. Reports from the six initial analysis teams and Phase-2 curation institutes are being announced individually as the results are published in scientific journals. After all the results of the initial analysis have been announced, an overall summary is planned.
Appendix
World’s first asteroid gas sample delivered by the Hayabusa2 mission:
A treasure box from Ryugu
1. Overview
Mass spectrometry and gas sampling were performed on the gas trapped in the sample container that was brought back to Earth by the Hayabusa2 spacecraft. Thirty hours after the re-entry capsule landed, the gas component within the sample container was extracted, sampled and examined with mass spectrometry using the GAs Extraction and Analysis equipment (GAEA) on site in Australia. After this, the gas sample was distributed to research institutes in Japan and overseas, and precise isotopic analysis of the gas components was performed. The results of this analysis revealed the gas contained a mixture of the solar wind and the Earth’s atmosphere. Based the amount of helium in the container, the trapped gas most likely originated in the solar wind that was liberated from the pulverisation of the surface of the grains in the Ryugu sample. The Hayabusa2 mission has become the first in the world to return gaseous components from a near-Earth orbit back to Earth.
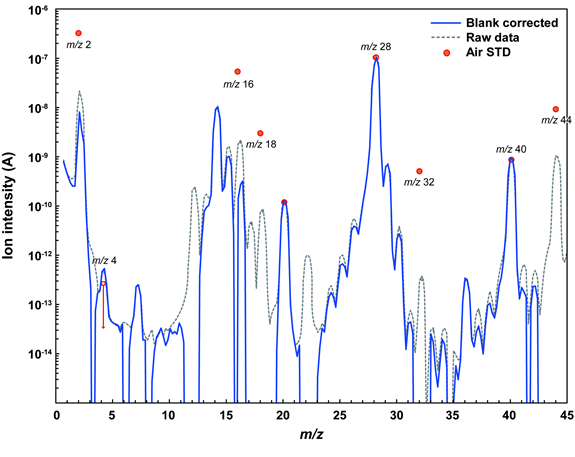
©Okazaki et al., 2022b
Figure 1: Mass spectrometry result of the gas extracted from the sample container obtained by the mass spectrometer attached to GAEA (solid blue line). The horizonal axis is the mass-to-charge ratio (m/z) and the vertical axis is the signal intensity of the ions at that m/z value (arbitrary scale). The blue line shows the signal once the contribution from the instrument itself (dotted grey line) has been subtracted. There is an excess of gas with an m/z of 4 (helium) compared to the Earth atmospheric gas (red circle).
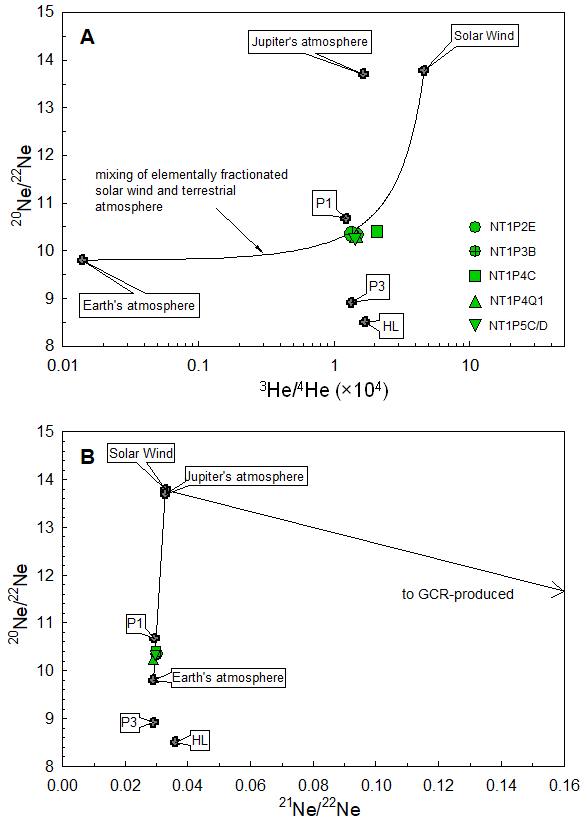
©Okazaki et al., 2022b
Figure 2: Helium and neon isotopic composition of the Hayabusa2 sample container gas (green). The composition can be explained by a mixture of the Earth’s atmosphere and the solar wind. Solid lines show the result of mixing the solar wind and Earth’s atmosphere in different ratios. Jupiter’s atmosphere, primordial noble gases (P1), pre-solar noble gases (P3, HL) and galactic cosmic-ray produced noble gases (GCR-produced) are shown for comparison.
2.Text
The Hayabusa2 spacecraft landed the re-entry capsule in the Woomera Prohibited Area of Australia on December 6, 2020. The re-entry capsule contained the sample container in which material from asteroid Ryugu had been collected by the spacecraft. The sample container was hermetically sealed with a metal seal mechanism. The gas component trapped in the sample container was analysed using mass spectrometry and sampled 30 hours after the capsule returned to Earth.
The re-entry capsule recovered from Woomera was initially brought to a research facility that had been established on-site (the Quick Look Facility). The capsule was then made safe and cleaned, before the sample container was removed from the capsule. The sample container was then connected to the vacuum equipment of the GAs Extraction and Analysis system (GAEA) that had been transported from Japan. While in vacuum conditions, a hole was opened in the bottom of the container which allowed the gas to pass through to GAEA without exposure to the atmosphere. According to GAEA’s vacuum pressure gauge, the internal pressure of the container was 68 Pa, which is less than 1/1000 of the Earth’s atmosphere, and two orders of magnitude lower than the gas pressure inside the sample container returned by Hayabusa. This demonstrated the effectiveness of the Hayabusa2 metal sealing system. However, the gas pressure was not expected to be purely from extra-terrestrial gas, but also limited terrestrial leakage from the Earth’s atmosphere.
After measuring the pressure, about 80% of the gas was collected in a special metal container. The remainder of the gas was passed to the mass spectrometer (Quadrupole Mass Spectrometer, QMS) that was mounted on GAEA. The main components of the gas were found to have a mass-to-charge ratio (m/z) of 2 (hydrogen molecules), 4 (helium-4), 28 (nitrogen molecules or carbon monoxide) and 40 (argon-40).
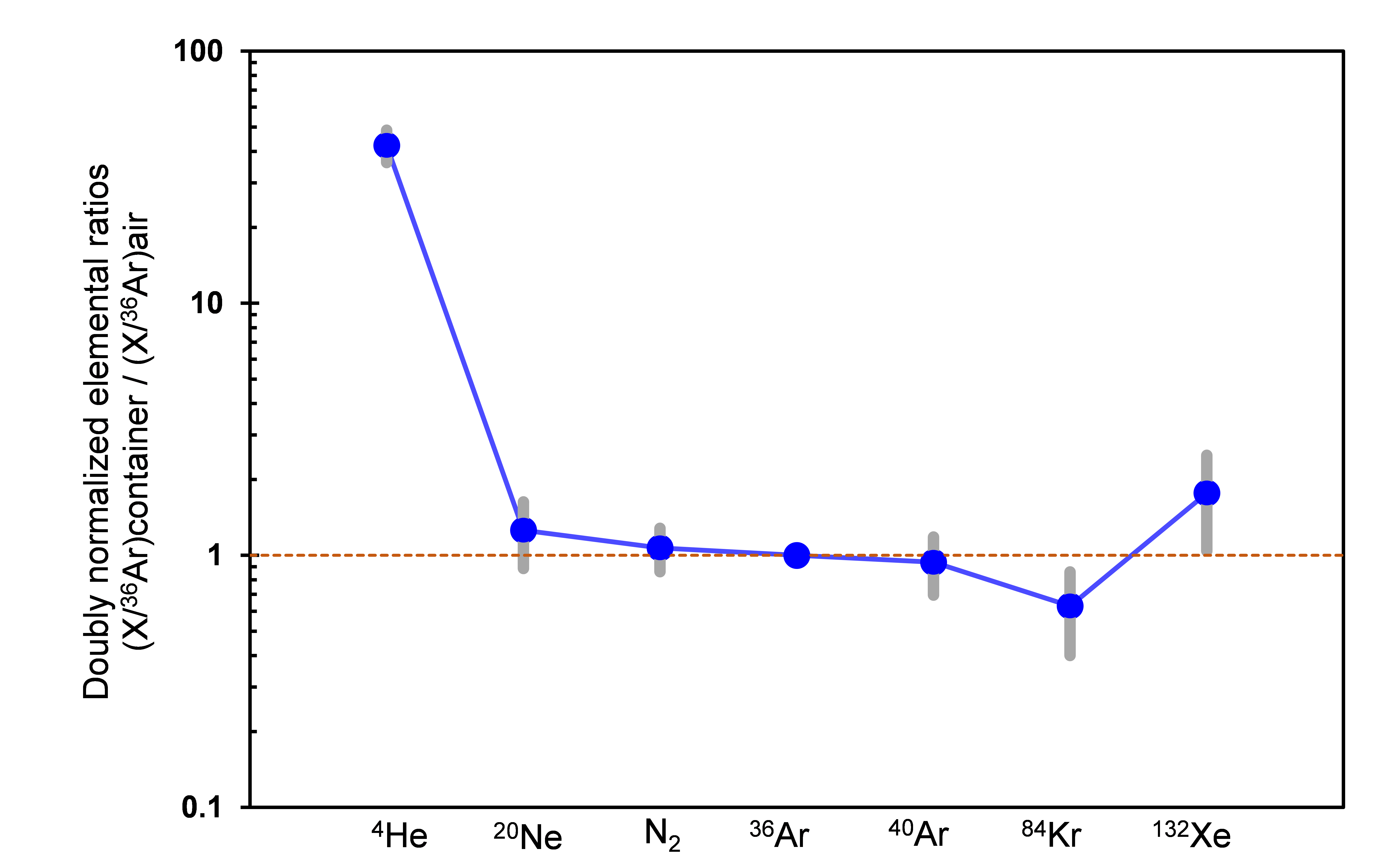
©Okazaki et al., 2022b
Figure 3: Elemental abundance of the sample container gas (solid blue line). Errors in the measurement are indicated by the grey bars. The vertical axis is the abundance normalized by 36Ar and the composition of the Earth’s atmosphere. The horizontal line indicates the gas species. Helium is in excess of one order of magnitude compared to the Earth’s atmosphere (orange dotted line).
To conduct a more detailed isotopic analysis, part of the extracted gas was collected in metal containers and distributed to seven research institutes in Japan and overseas. These analyses found that the isotope ratio of helium with an atomic mass of 3 (3He) is 100 times higher than in the Earth’s atmosphere (Figure 2). The isotope composition of neon also differed from that of the Earth’s atmosphere. From examining the elemental abundance ratios of helium, neon, and argon, and the helium isotope ratio in the container, it was discovered that the container gas composition could be explained with a mix of solar wind and the Earth’s atmosphere.
Conversely, the gas with m/z 28 was found to be molecular nitrogen, and the isotopic composition was discovered to be close to that found in the Earth’s atmosphere. The isotopic composition of argon was also found to be close to that of the Earth’s atmosphere. This indicates that most of the gases, other than helium and neon, originate from the Earth’s atmosphere (Figure 3). Additionally, water (as a vapour) and methane gas, the former of which may come out from hydrated silicates, were not detected even in very highly sensitive analysis. This suggests that the water in the hydrated silicates in the Ryugu sample remained firmly bound to the sample grains.
The release of the solar wind-derived helium and neon from the Ryugu sample was examined based on the amount of helium in the sample container, the recovered mass of the Ryugu sample grains, the grain size distribution, and the helium gas concentration. Solar wind gas can be trapped by grains at the surface of the asteroid and released if those grains fragment within the sample container during spacecraft operations. The expected efficiency of solar wind-derived helium release was calculated from two possible fracture modes of the sample grains (figure 4): “isotropic fragmentation” of the Ryugu sample grain within the container, and “surface pulverisation” of only the surface of the sample grain that was exposed to the solar wind. The results from this found that the amount of helium in the container can be explained by surface pulverisation if approximately 2% of the total surface area of each grain fragments. In the case of the isotropic fragmentation, the sample grains must be crushed into fine particles. This is less likely, as the material found in the Ryugu sample catcher contained larger particles several millimetres in size. The possibility of heating during re-entry through the atmosphere was also considered, but the temperature of the sample container did not exceed 65°C, so it can be assumed that the thermal effects on the Ryugu sample were small.
The gas trapped in the container proved that Hayabusa2 was able to not only bring solid material from asteroid Ryugu back to Earth, but also gas components released by the sample. This makes the Hayabusa2 mission the first in the world to collect and bring back a gas sample from such a near-Earth orbiting object. The sample container was therefore truly a “treasure box” from Ryugu.
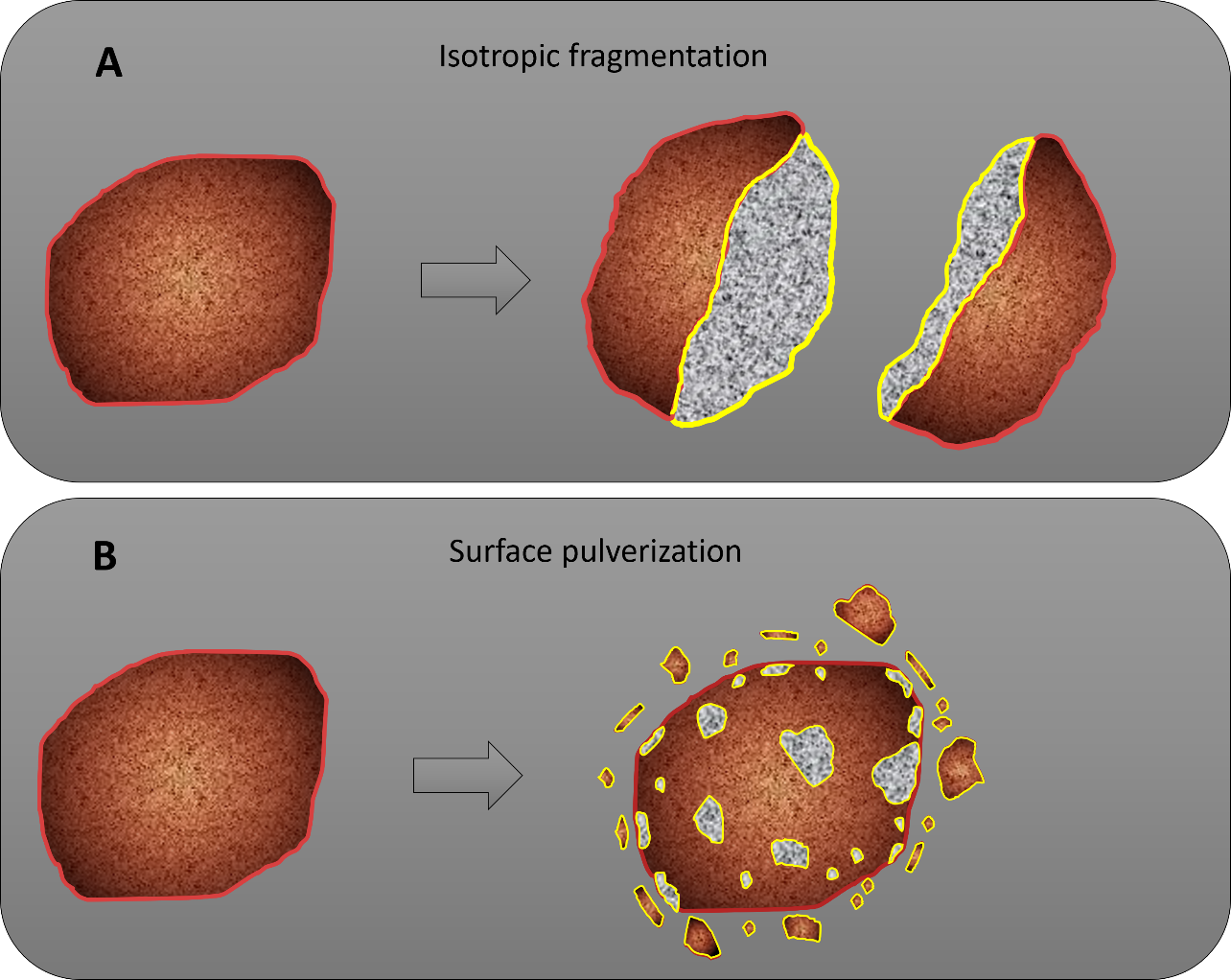
©Okazaki et al., 2022b
Figure 4: Possible modes of fragmentation for the Ryugu grains within the sample container. (A) In the case of isotopic fragmentation, the solar wind is released from only a small area (yellow line). (B) When the grain surface is pulverised, the original particle size does not change significantly, but the surface that releases the solar wind (yellow line) is much larger.
3. Publication information
Journal Title: Science Advances
DOI: 10.1126/sciadv.abo7239
Title of paper: First asteroid gas sample delivered by the Hayabusa2 mission: A treasure box from Ryugu
Authors: R. Okazaki1, Y.N. Miura2, Y. Takano3, H. Sawada4, K. Sakamoto4, T. Yada4, K. Yamada5, S. Kawagucci6,7, Y. Matsui6,7, K. Hashizume8, A. Ishida9, M.W. Broadley10, B. Marty10, D. Byrne10, E. Füri10, A. Meshik11, O. Pravdivtseva11, H. Busemann12, M.E.I. Riebe12, J. Gilmour13, J. Park14,15, K. Bajo16, K. Righter17, S. Sakai3, S. Sekimoto18, F. Kitajima1, S.A. Crowther13, N. Iwata19, N. Shirai20,21, M. Ebihara20, R. Yokochi22, K. Nishiizumi23, K. Nagao24, J.I. Lee24, P. Clay13, A. Kano25, M.W. Caffee26,27, R. Uemura28, M. Inagaki18, D. Krietsch12, C. Maden12, M. Yamamoto1, L. Fawcett13, T. Lawton13, T. Nakamura9, H. Naraoka1, T. Noguchi29, H. Yabuta30, H. Yurimoto16, Y. Tsuda4, S. Watanabe28, M. Abe4,31, M. Arakawa32, A. Fujii4, M. Hayakawa4, N. Hirata32, N. Hirata33, R. Honda34, C. Honda33, S. Hosoda4, Y. Iijima4†, H. Ikeda4, M. Ishiguro35, Y. Ishihara36, T. Iwata4, K. Kawahara4, S. Kikuchi37,38, K. Kitazato33, K. Matsumoto31,38, M. Matsuoka39, T. Michikami40, Y. Mimasu4, A. Miura4, T. Morota25, SatS.oru Nakazawa4, N. Namiki31,38, H. oda31,38, R. Noguchi41, N. Ogawa4, K. Ogawa36, T. Okada4,42, C. Okamoto32†, G. Ono43, M. Ozaki4, 31, T. Saiki4,31, N. Sakatani44, H. Senshu37, Y. Shimaki4, K. Shirai4,32, S. Sugita25, Y. Takei4, H. Takeuchi4, S. Tanaka4, E. Tatsumi25,45, F. Terui46, R. Tsukizaki4, K. Wada37, M. Yamada37, T. Yamada4, Y. Yamamoto4, H. Yano4,31, Y. Yokota4, K. Yoshihara4, M. Yoshikawa4,31, K. Yoshikawa4, S. Furuya4, K. Hatakeda47, T. Hayashi4, Y. Hitomi47, K. Kumagai47, A. Miyazaki4, A. Nakato4, M. Nishimura4, H. Soejima47, A. Iwamae47, D. Yamamoto4,48, K. Yogata4, M. Yoshitake4, R. Fukai4, T. Usui4, T. Ireland49, H.C. Connolly Jr.50. D.S. Lauretta51, S. Tachibana52
1Department of Earth and Planetary Sciences, Kyushu University, Fukuoka, 819-0395, Japan.
2Earthquake Research Institute, The University of Tokyo, Tokyo, 113-0032, Japan.
3Biogeochemistry Research Center, Japan Agency for Marine-Earth Science and Technology (JAMSTEC), Yokosuka, Kanagawa, 237-0061, Japan.
4Institute of Space and Astronautical Science, Japan Aerospace Exploration Agency (JAXA), Sagamihara, 252-5210, Japan.
5Department of Chemical Science and Engineering, Tokyo Institute of Technology, Yokohama, Kanagawa, 226-8503, Japan.
6Research Institute for Global Change, JAMSTEC, Yokosuka, 237-0061, Japan.
7Institute for Extra-cutting-edge Science and Technology Avant-garde Research (X-star), JAMSTEC, Yokosuka, 237-0061, Japan.
8Faculty of Science, Ibaraki University, Mito, 310-8512, Japan.
9Department of Earth Science, Tohoku University, Sendai, 980-8578, Japan.
10Université de Lorraine, CNRS, CRPG, F-54000 Nancy, France.
11Physics Department, Washington University, Saint Louis, MO, 63130, USA.
12Institute of Geochemistry and Petrology, Eidgenössische Technische Hochschule (ETH) Zürich, 8092 Zürich, Switzerland.
13Department of Earth and Environmental Sciences, The University of Manchester, Manchester, M13 9PL, UK.
14Physical Sciences, Kingsborough Community College, The City University of New York, Brooklyn, NY, 11235, USA.
15Department of Earth and Planetary Sciences, American Museum of Natural History, NY, 10024, USA.
16Department of Earth and Planetary Sciences, Hokkaido University, Sapporo, 060-0810, Japan.
17Astromaterials Research and Exploration Science, Mailcode XI2, National Aeronautics and Space Administration (NASA) Johnson Space Center, Houston, TX, 77058, USA.
18Institute for Integrated Radiation and Nuclear Science, Kyoto University, Osaka, 590-0494, Japan.
19Faculty of Science, Yamagata University, Yamagata, 990-8560, Japan.
20Graduate School of Science and Engineering, Tokyo Metropolitan University, Hachioji, Tokyo, 192-0397, Japan.
21Department of Chemistry, Faculty of Science, Kanagawa University, Hiratsuka, Kanagawa, 259-1293, Japan.
22Department of the Geophysical Sciences, The University of Chicago, Chicago IL, USA.
23Space Sciences Laboratory, University of California, Berkeley, CA, 94720, USA.
24Division of Earth Sciences, Korea Polar Research Institute, Incheon, 21990, Korea.
25School of Science, The University of Tokyo, Tokyo, 113-0033, Japan.
26Department of Physics and Astronomy, Purdue University, West Lafayette, IN 47907, USA.
27Department of Earth, Atmospheric, and Planetary Sciences, Purdue University, West Lafayette, IN 47907, USA.
28Department of Earth and Environmental Sciences, Nagoya University, Nagoya, 464-8601, Japan.
29Division of Earth and Planetary Sciences, Kyoto University, Kyoto, 606-8502, Japan.
30Department of Earth and Planetary Systems Science, Hiroshima University, Higashi-Hiroshima, 739-8526, Japan.
31Department of Space and Astronautical Science, The Graduate University for Advanced Studies, Hayama 240-0193, Japan.
32Department of Planetology, Kobe University, Kobe, 657-8501, Japan.
33Aizu Research Cluster for Space Science, University of Aizu, Aizu-Wakamatsu, 965-8580, Japan.
34Center of Data Science, Ehime University, Matsuyama, 790-8577, Japan.
35Department of Physics and Astronomy, Seoul National University, Seoul, 08826, Republic of Korea.
36JAXA Space Exploration Center, JAXA, Sagamihara, 252-5210, Japan.
37Planetary Exploration Research Center, Chiba Institute of Technology, Narashino, 275-0016, Japan.
38National Astronomical Observatory of Japan, Mitaka, 181-8588, Japan.
39Geological Survey of Japan, National Institute of Advanced Industrial Science and Technology, Ibaraki, 305-8567, Japan.
40Faculty of Engineering, Kindai University, Higashi-Hiroshima, 739-2116, Japan.
41Faculty of Science, Niigata University, Niigata, 950-2181, Japan.
42Department of Chemistry, The University of Tokyo, Tokyo 113-0033, Japan.
43Research and Development Directorate, JAXA, Sagamihara, 252-5210, Japan.
44Department of Physics, Rikkyo University, Tokyo, 171-8501, Japan.
45Instituto de Astrofísica de Canarias, University of La Laguna, Tenerife, Spain.
46Department of Mechanical Engineering, Kanagawa Institute of Technology, Atsugi, 243-0292, Japan.
47Marine Works Japan Ltd., Yokosuka, 237-0063, Japan.
48Department of Earth and Planetary Science, Tokyo Institute of Technology, Ookayama, Tokyo, 152-8550, Japan.
49School of Earth and Environmental Sciences, The University of Queensland, St Lucia QLD, 4072, Australia.
50Department of Geology, School of Earth and Environment, Rowan University, Glassboro, NJ, 08028, USA.
51Lunar and Planetary Laboratory, University of Arizona, Tucson, AZ, 85721, USA.
52UTokyo Organization for Planetary and Space Science, The University of Tokyo, Tokyo, 113-0033, Japan.
†Deceased.